More than just a simple push...
In part 1 – we discussed the advantages of transdermal drug delivery. Part 2 looked at ways to make molecules more efficient at skin penetration. In this section, we will cover patch technology and how they help facilitate drug delivery.
Transdermal patches contain a backing layer and an adhesive layer. Wrapped inside those layers is the technology that allows for drug delivery. Very simply – patches can be categorized into passive patches and active patches.
Most passive patches contain a reservoir, matrix, or drug-in-adhesive (DIA) formulation. Once these types of patches are applied to the skin, the release of medication is regulated by membranes inside the patch, body temperature of the skin and other permeation enhancers. As we discussed in previous posts, some molecules are ideal for passive permeation. Drugs such as scopolamine, lidocaine, and rivastigmine, because of a favorable permeation profile have had excellent commercial success.
For active patches – the three main areas of innovation are: electroporation, microneedle, and iontophoresis.
Electroporation uses short (millisecond), high-voltage pulses to disrupt the stratum corneum. This creates ‘electropores’ in outer layer of the skin that can persist for hours – which is favorable to the passage of large molecules such as peptides, vaccines, and DNA. Although electroporation has been studied extensively in animals, this approach to transdermal delivery has limited attention in humans due largely to the complexity of device design1.
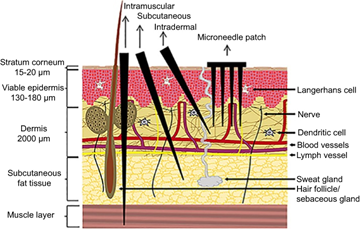
Fig 1
Microneedles are applied on the skin as a patch. When pressed to the surface of the skin, these needles create microscopic pores, which allows the drug to enter the body. Due to the nearly microscopic size of these microneedles, dermal nerves and blood vessels are not affected, so there is no pain or bleeding when the patch is applied.
Transdermal drug delivery for disease treatment is a key application for microneedles. Conditions such as cancer, diabetes and obesity have extensive development and commercialization of microneedle applications. Other applications for microneedles include immunoglobulin delivery, disease diagnosis, and cosmetic applications3.
Iontophoresis is a non-invasive method of delivering medication through the skin using a low level electrical current. Iontophoresis works on the concept of ‘similar charges repel each other’. A water soluble molecule is placed on the similarly charged pad. The opposite pad has saline applied to complete the circuit. When the patch is applied – the active molecule is REPELLED away from the charged pad and through the skin into the viable epidermis and dermis.
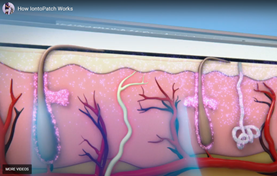
To see how this works, click here:
The active molecule – depending on penetration can act locally or be slowly absorbed into systemic circulation. Aside from being non-invasive, iontophoresis allows for a controlled and sustained delivery of medication over the prescribed period of time.
Iontophoresis has been used for decades - mostly commonly in sports medicine and orthopedic surgery to treat post operative pain and to deliver anti- inflammatory medication to treat injuries such as tennis elbow, plantar fasciitis, Achilles tendinitis and rotator cuff injuries.
To make things simpler and more convenient for the patient, slim and flexible iontophoresis patches have been developed so patients can easily self-administer their medication at home, discretely, and without interfering with their lifestyle.
In this series, we have discussed:
- The advantages of transdermal drug delivery
- Ways to make the molecules themselves more favorable for skin penetration
- How patches are designed to deliver those molecules more effectively
In the final edition of this series, we will look at what happens when science takes these enhanced molecules and combines them with active patch delivery.
- Prausnitz M., and Langer, R., Nat Biotechnol. 2008 Nov; 26(11): 1261–1268. doi: 10.1038/nbt.1504
- Fig 1: Reproduced from Leone M, Mönkäre J, Bouwstra JA, Kersten GF. Dissolving Microneedle Patches for Dermal Vaccination. Pharm Res. 2017;34(11):2223-2240
- Yang J., Acta Pharmaceutica Sinica B, 2019; 9(3):469-483 https://doi.org/10.1016/j.apsb.2019.03.007